
Artificial Intelligence for early cancer detection in Nature Medicine
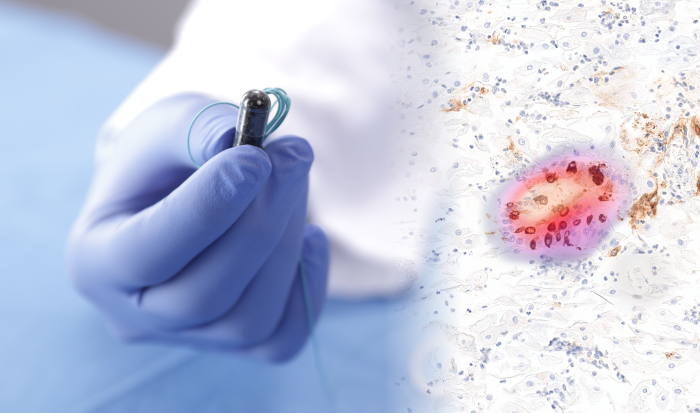
CAMBRIDGE, UK, 15 April 2021: Dr Marcel Gehrung, CEO of Cyted, was part of the research group that developed a new technique to help experts diagnose Barrett’s oesophagus – a pre-cancerous condition that can increase the risk of developing oesophageal cancer.
Published todayinNature Medicineand funded by Cancer Research UK, the study explored how artificial intelligence could help free up pathologists time and allow them on diagnosing the trickiest cases of Barrett’s oesophagus.
Cytosponge is a new diagnostic tool developed by Cancer Research UK scientists at the University of Cambridge. It uses a ‘sponge on a string’ to collect cells from the oesophagus, which are then sent to the lab for testing, where pathologists look for a biomarker linked to Barrett’s oesophagus, TFF3.
Previous researchhas suggested that the Cytosponge-TFF3 test can identify 10 times more people with Barrett’s oesophagus than current GP care. However, if this test were to be more commonly used in GP surgeries and elsewhere within the NHS, it could increase the demand on NHS services.
Barrett’s oesophagus can cause cells in the oesophagus to grow abnormally, increasing the risk of oesophageal cancer. Around 3 to 13% of people with the Barrett’s oesophagus develop a type of oesophageal cancer called oesophageal adenocarcinoma –11 times more than the average person. It is thought that many cases of Barrett’s oesophagus go undetected.
The work carried out by Dr Marcel Gehrung during his PhD, based at the Cancer Research UK Cambridge Institute, led to the founding of Cyted.
Under the supervision of Dr Florian Markowetz at Cancer Research UK Cambridge Institute the team wanted to see if AI could help ease the burden of analysing samples, paving the way for Cytosponge to be used more frequently without straining an already busy NHS care pathway.
Dr Marcel Gehrung, CEO of Cyted, commented:
“A major bottleneck for scaling Cytosponge to test large patient populations is the time it takes for a pathologist to analyse the samples, which has several time-consuming steps.”
The researchers developed an approach that applied deep learning (an AI function that tries to mimic the workings of the brain and process raw data without human oversight) to Cytosponge samples taken from patients taking part in clinical trials. Images from these samples were analysed by the model and trained to understand the features of particular cells that indicate the presence of Barrett’s oesophagus, called goblet cells.
Coding a pathologist’s partner
Trying to develop a fully-automated system with the ability to mimic a pathologist’s single “positive” or “negative” diagnosis was complex. From the samples taken from 2,331 patients, pathologists were able to correctly identify 82% of Barrett’s oesophagus cases. In comparison, the AI approach was able to correctly identify 73% of cases, with both approaches correctly identifying 93% of the negative cases.
Instead, researchers were guided by experienced pathologists to develop a semi-automated triage system. This meant that the result for each sample was categorised into one of 8 different classes depending on how clear cut the diagnosis and quality was. Samples that were deemed to be low quality or more challenging for the deep-learning model were assessed manually by pathologists.
This triage system proved successful. When applied to the clearer cut cases (approximately 60% of the samples), the algorithm was able to identify 83% of cases.
Researchers suggest that it could reduce Cytosponge-related workload for pathologists by 57%.
The future of AI
Michelle Mitchell, chief executive of Cancer Research UK, said that pathologists play a key role in diagnosis. “But like so many other areas of the NHS, they have been seriously impacted by a lack of investment in workforce over the years. Research such as this, exploring how to support pathologists in their vital work through new technology and innovations is vital, as is long term investment and planning of the cancer workforce.”
As the use and development of this deep learning tool continues to evolve, the researchers think that it will become even more accurate in its definition of triage classes. Researchers also say the technology isn’t limited to this one condition and in the future, it could potentially be applied to other conditions including pancreatic, thyroid and bowel cancer.
References
- Gehrung et al 2021 Triage-driven diagnosis of Barrett esophagus for early detection of esophageal adenocarcinoma using deep learning
- Fitzgerald et al 2020 Cytosponge-trefoil factor 3 versus usual care to identify Barrett's oesophagus in a primary care setting: a multicentre, pragmatic, randomised controlled trial
- Oesophageal cancer risk | Cancer Research UK
About Cyted
Cyted is enabling the earlier detection of disease, providing products and services that combine proprietary artificial intelligence technology and digital diagnostic infrastructure with next generation biomarkers.
With significant expertise in pathology, the Company is developing a pipeline of digital and molecular tests for disease detection and risk stratification to streamline diagnostic and clinical workflows. Cyted has the capacity to rapidly implement its sample processing and analysis workflow at scale via Dialogical®, its proprietary pathology reporting platform based on machine learning algorithms.
The Company’s services and products are initially focused on oesophageal cancer, where the combination of an identifiable at-risk population coupled with excellent therapy options offers a unique opportunity to improve patient outcomes through early detection.
